Learn More
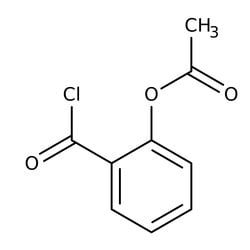
O-Acetylsalicyloylchlorid, 97 %, Thermo Scientific Chemicals
Description
O-Acetylsalicyloyl chloride was used in the synthesis of enantiomerically pure bidentate heteroorganic ligands built on simple achiral skeletons and containing an aziridine moiety. It was used in the general synthesis of acetoxybenzamides. It was used in the chemical synthesis of 2,2,6,6-tetramethyl-1-piperidinyloxy(TEMPO)-aspirin conjugate via condensation reaction with 4-hydroxy-TEMPO. It was used as reagent in acylation of cyclobutenediones.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
O-Acetylsalicyloylchlorid wurde bei der Synthese von enantiomerisch reinen heteroorganischen Bidentat-Liganden verwendet, die auf einfachen achiralen Skeletten gebaut wurden und eine Aziridingruppe enthielten. Es wurde bei der allgemeinen Synthese von Acetoxybenzamiden verwendet. Es wurde bei der chemischen Synthese von 2,2,6,6-Tetramethyl-1-Piperidinyloxy(TEMPO)-Aspirin-Konjugat über Kondensationsreaktion mit 4-Hydroxy-TEMPO verwendet. Es wurde als Reagenz bei der Acylierung von Cyclobutendionen verwendet.
Löslichkeit
Löslich in Toluol. Es reagiert mit Wasser.
Hinweise
Feuchtigkeitsempfindlich. Unter trockenem Inertgas lagern. Nicht in der Nähe starker Basen aufbewahren.

Specifications
Specifications
| Chemischer Name oder Material | O-Acetylsalicyloyl chloride |
| Schmelzpunkt | 47°C to 50°C |
| Siedepunkt | 134°C to 136°C (12 mmHg) |
| Flammpunkt | >110°C (230°F) |
| Geruch | Pungent, Makes Eyes Water |
| Menge | 5 g |
| UN-Nummer | UN3261 |
| Beilstein | 880372 |
| Formelmasse | 198.61 |
| Reinheit (%) | 97% |
RUO – Research Use Only
By clicking Submit, you acknowledge that you may be contacted by Fisher Scientific in regards to the feedback you have provided in this form. We will not share your information for any other purposes. All contact information provided shall also be maintained in accordance with our Privacy Policy.